IMLS Update: Removing Copper Corrosion
| Written by | Jessica Lafrance-Hwang |
|---|---|
| Published | 7/17/2015 |
IMLS Update: Removing Copper Corrosion
| Written by | Jessica Lafrance-Hwang |
|---|---|
| Published | 7/17/2015 |

Our IMLS Grant Conservation staff uses scientific and aesthetic training to conserve, clean and repair a large number of Communications collections. A familiar problem we often encounter is copper “rust” that disfigures objects. Conservators call these damages “corrosion products”. The corrosion is actually “eating” the metal as it forms on a range of object types. Copper corrosion products form on copper and copper alloys (like brass) through chemical reactions that are initiated by contact with various materials nearby and from the air pollution. Nearby materials that corrosion include fatty acids in waxes and leather dressing, sulfur in rubber products, or salts in water or human sweat. Copper corrosion products vary greatly. They can be very waxy or hard and mineralized or soft and powdery, depending on what caused it.


In photo above you can see bright green waxy copper corrosion product on the brass plate of a telephone handle. This was caused by a thin piece of leather that had been glued to the plate. Over time the organic acids in the oils and waxes applied to the leather caused the corrosion to occur. Waxy corrosion products are easy but time consuming to remove using mineral spirits, swabs, and sharpened wooden tools.


In this image you can see yellow copper corrosion products caused on screws covered by small hard-rubber gaskets inside a typewriter. Sulfur in the rubber can undergo reactions to become sulfuric acid which can then cause dramatic and very mineral-looking corrosion products to form. The corrosion products are often removed using a scalpel or dental tool, while being very cautious of the often deteriorated rubber nearby.
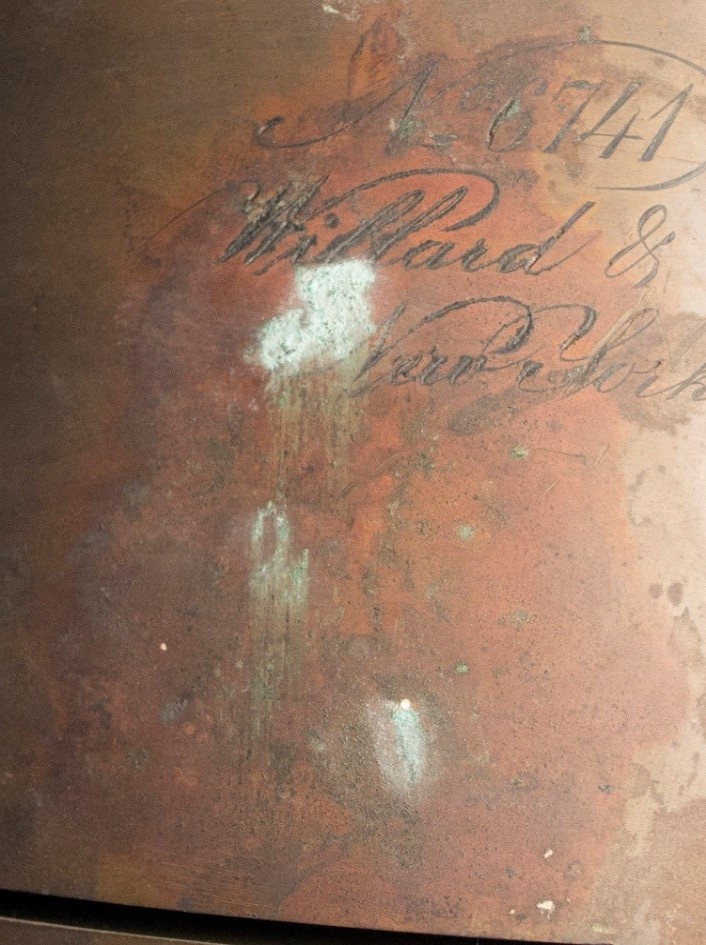

In the photo aboive you can see soft powdery light colored corrosion products on the tarnished surface of a large portrait camera lens. The salts and oils that are naturally in our sweat and on our hands transfer to the surface of objects when handled without gloves. Over time these salty deposits can become centers for corrosion to start. This type of corrosion can be difficult to remove as it is soft and powdery on the surface but eats into the metal and creates a small pit. Conservators in the lab often used fiberglass brushes, scalpels, and sometimes chemical solutions to remove the corrosion.
Removing the corrosion doesn’t mean it won’t happen again. The proper environment (low humidity!) and proper handling need to be used along with conservation work to minimize corrosion in storage.
Read more about our IMLS work to date:
- Behind the Scenes with IMLS: The Process
- Cadmium in Collections
- A Peek “Behind the Scenes” at The Henry Ford
- Just Added to Our Digital Collections: Atari Burial Collection
- Just Added to Our Digital Collections: Insulators
- Just Added to Our Digital Collections: IMLS Grant Artifacts
Jessica Lafrance-Hwang is Project Conservator, IMLS Communications Grant Project, at The Henry Ford.
Keywords |
|---|
