Posts Tagged by louise stewart beck
Imagine this: the Institute of Museum and Library Services (IMLS) project team is working in the Collections Storage Building, selecting objects to be conserved as part of our grant-funded work. From the top level of pallet racking, about 15 feet above the ground, we remove some pallets of boxes and bring them down to ground level to unpack. We then climb the moveable stairs to take a peek at the area that we have exposed. The sight that greets us is confusing, but intriguing: a giant, golden-toned teapot, sitting in the center of the racking, far enough back that it was not visible from the ground. It was almost like revealing a magic lamp! We test-lifted it and realized that it was very light for its size, and must be hollow, so we carefully moved it off of the racking and to ground level

The giant teapot trade sign as we found it in the Collections Storage Building (after we had moved it down from the top shelf).
From the bracket that we found on the handle, it quickly became apparent that this was some sort of a trade sign, likely for a tea shop or coffee house. The body of the teapot occupies a space about three feet on every side – it would have been a very eye-catching sign! A little bit of research led us to some other interesting examples, including one that currently hangs above a Starbucks in Boston and is set up to blow steam out of its spout!
Our teapot has some mysteries, though – the golden paint has some texture to it, as if there were at one point a stripe along the widest part of the teapot’s body, with vertical stripes reaching from that stripe to the lid. Was the teapot originally painted a different color, or with a pattern? We did some minor tests to see if we could isolate different layers of paint, but we were not successful. We might decide in the future to do a more thorough analysis, but that would be after discussion with the curators. We also noted that our giant teapot does not have a hollow spout, and therefore, despite being hollow, probably never had the mechanism to blow steam in the same way as some others.

The giant teapot on the table in the lab - you can really get a sense of how large it is!
Ultimately, we don’t know a lot about where the giant teapot was originally used, or where it may be displayed in the future. We treated this object with nothing more than a simple cleaning – it was overall very stable to begin with, just dusty and dirty from being in storage. By minimizing treatment to the point of only stabilizing the object, we are leaving the option open for a future conservator to do more work while still ensuring that it’s going to be safe and sound in storage. It also allows us to treat more objects from storage as we progress through the grant. Maybe someday in the future we’ll see the giant teapot again, but for now it’s safe and sound in the Main Storage Building! You can check it out in our Digital Collections.

The giant teapot after treatment, ready to go back to storage.
Louise Stewart Beck is Senior Conservator at The Henry Ford.
IMLS grant, conservation, collections care, by Louise Stewart Beck, #Behind The Scenes @ The Henry Ford
IMLS Update: Analysis and New Tools
 This blog post is part of a series about storage relocation and improvements that we are able to undertake thanks to a grant from the Institute of Museum and Library Services.
This blog post is part of a series about storage relocation and improvements that we are able to undertake thanks to a grant from the Institute of Museum and Library Services.
In the course of our work as conservators, we get some very exciting opportunities. Thanks to a partnership with Hitachi High Technologies, for the past few months the conservation lab here at The Henry Ford has had a Scanning Electron Microscope (SEM) with an energy-dispersive x-ray (EDX) spectroscopy attachment in our lab.
 What does this mean? It means that not only have we been able to look at samples at huge magnifications, but we have had the ability to do elemental analysis of materials on-demand. Scanning electron microscopy uses a beam of electrons, rather than light as in optical microscopes, to investigate the surface of sample. A tungsten filament generates electrons, which are accelerated, condensed, and focused on the sample in a chamber under vacuum. There are three kinds of interactions between the beam and that sample that provide us with the information we are interested in. First, there are secondary electrons – the electron beam hits an electron in the sample, causing it to “bounce back” at the detector. These give us a 3D topographical map of the surface of the sample. Second, there are back-scattered electrons – the electron beam misses any electrons in the sample and is drawn towards a positively-charged nucleus instead. The electrons essentially orbit the nucleus, entering and then leaving the sample quickly. The heavier the nucleus, the higher that element is on the periodic table, the more electrons will be attracted to it. From this, we get a qualitative elemental map of the surface, with heavier elements appearing brighter, and lighter elements appearing darker.
What does this mean? It means that not only have we been able to look at samples at huge magnifications, but we have had the ability to do elemental analysis of materials on-demand. Scanning electron microscopy uses a beam of electrons, rather than light as in optical microscopes, to investigate the surface of sample. A tungsten filament generates electrons, which are accelerated, condensed, and focused on the sample in a chamber under vacuum. There are three kinds of interactions between the beam and that sample that provide us with the information we are interested in. First, there are secondary electrons – the electron beam hits an electron in the sample, causing it to “bounce back” at the detector. These give us a 3D topographical map of the surface of the sample. Second, there are back-scattered electrons – the electron beam misses any electrons in the sample and is drawn towards a positively-charged nucleus instead. The electrons essentially orbit the nucleus, entering and then leaving the sample quickly. The heavier the nucleus, the higher that element is on the periodic table, the more electrons will be attracted to it. From this, we get a qualitative elemental map of the surface, with heavier elements appearing brighter, and lighter elements appearing darker.  Conservation Specialist Ellen Seidell demonstrates the SEM with Henry Ford Museum of American Innovation volunteer Pete Caldwell.
Conservation Specialist Ellen Seidell demonstrates the SEM with Henry Ford Museum of American Innovation volunteer Pete Caldwell.
The EDX attachment to the SEM allows us to go one step further, to a third source of information. When the secondary electrons leave the sample, they leave a hole in the element’s valence shell that must be filled. An electron from a higher valence shell falls to fill it, releasing a characteristic x-ray as it does so – the detector then uses these to create a quantitative elemental map of the surface. A ‘K’ from a stamp block, as viewed in the scanning electron microscope.
A ‘K’ from a stamp block, as viewed in the scanning electron microscope.
The understanding of materials is fundamental to conservation. Before we begin working on any treatment, we use our knowledge, experience, and analytical tools such as microscopy or chemical tests to make determinations about what artifacts are made of, and from there decide on the best methods of treatment. Sometimes, materials such as metal can be difficult to positively identify, especially when they are degrading, and that is where the SEM-EDX shines. Take for example the stamp-block letter shown here. The letter was only about a quarter inch tall, and from visual inspection, it was difficult to tell if the block was made of lead (with minor corrosion) or from heavily-degraded rubber. By putting this into the SEM, it was possible a good image of the surface and also to run an elemental analysis that confirmed that it was made of lead. Knowing this, it was coated to prevent future corrosion and to make it safe to handle.
Elemental analysis is also useful when it comes to traces of chemicals left on artifacts. We recently came across a number of early pesticide applicators, which if unused would be harmless. However, early pesticides frequently contained arsenic, so our immediate concern was that they were contaminated. We were able to take a sample of surface dirt from one of the applicators and analyze it in the SEM.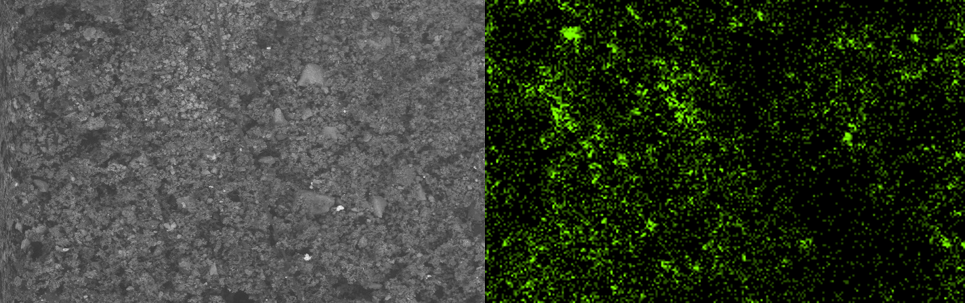 An SEM image of a dirt sample from an artifact (left) and a map of arsenic within that sample (right).
An SEM image of a dirt sample from an artifact (left) and a map of arsenic within that sample (right).
The image on the left is the SEM image of the dirt particles, and the image on the right is the EDX map of the locations of arsenic within the sample. Now that we know they are contaminated, we can treat them in a way that protects us as well as making the objects safe for future handling.
We have also used the SEM-EDX to analyze corrosion products, to look at metal structures, and even to analyze some of the products that we use to clean and repair artifacts. It has been a great experience for us, and we’re very thankful to Hitachi for the opportunity and to the IMLS as always for their continued support.
Louise Stewart Beck is the project conservator for The Henry Ford's IMLS storage improvement grant.
philanthropy, technology, IMLS grant, conservation, collections care, by Louise Stewart Beck, #Behind The Scenes @ The Henry Ford
Behind the Scenes with IMLS: Looking Back & Moving Forward
 As we start a new year, it’s a good time to look back on the first half of The Henry Ford’s IMLS-funded grant to work on electrical objects, and to take stock for the future.
As we start a new year, it’s a good time to look back on the first half of The Henry Ford’s IMLS-funded grant to work on electrical objects, and to take stock for the future.
We had a busy and productive fall 2016, with some new adventures thrown in with continuing progress on objects themselves. If you haven’t already seen them, you should check out our Facebook Live videos – we’ve done a few so far (in October, November, and December), and the plan is to continue doing them on the first Friday of each month.
 Gaulard & Gibbs transformer on the shelf before treatment (29.1333.229).
Gaulard & Gibbs transformer on the shelf before treatment (29.1333.229).
This Gaulard & Gibbs transformer had several conservation issues when we first saw it, most notably that the wooden base had broken under the strain of the weight of the object itself. You can see this in the before picture, where the object is lying on its side because it cannot stand anymore. There are also faint hints of color along the metal tabs that run up the body of the object. The Gaulard and Gibbs transformer after treatment (29.1333.229).
The Gaulard and Gibbs transformer after treatment (29.1333.229).
You can see that this transformer had a fantastic transformation during conservation treatment – simply removing years of built-up dust revealed a very vivid red and black coloration. The broken wooden base was also very successfully repaired, and it is now possible for the object to stand on its feet again. When it’s packed for storage, it will be lying down again, so that the weakened wooden base isn’t put under too much strain for long periods of time.
We featured this object briefly in our Facebook Live videos – you may have noticed, if you tuned into both, that you could see the ‘before’ and ‘after’ as they happened.
The interior of a meter, with strange accretions on white enameled metal. Note that this view is of the reverse of the top face (29.1333.63)
We’ve also encountered some interesting materials and material problems in the first half of our IMLS grant work. One of the most interesting was this strange accretion, found on the interior of a meter. Those brownish bulbs appeared to be seeping into the object from the top, but were only present on the enameled portions of the metal. They were friable and lighter on the inside than the outside. We looked at samples under the microscope, and even attempted to culture a sample, in case it’s a type of mold (it does not appear to be). We’re still not sure what exactly they are, but we will continue to try to figure it out! Mysteries of the museum, indeed. An ohmmeter with a great example of hard rubber – note that the cylindrical casing which would usually go over the black area is removed in this photo (31.1217.235)
An ohmmeter with a great example of hard rubber – note that the cylindrical casing which would usually go over the black area is removed in this photo (31.1217.235)
We have also recently come across a fantastic example of perfectly preserved hard rubber. The base of the object is one solid slab of hard rubber, but the protected interior area has retained the original black, mirror-like finish. The discoloration and matte surface of hard rubber occurs primarily from light exposure over time, and the colors possible range from a light black to the red-brown color on this object. We’ve put the exterior cylindrical case back on the object, sealing it well, so that the very tight case can continue to preserve this fantastic interior.  Conservator Cuong Nguyen and Conservation Technician Andrew Ganem working on motors in their lab.
Conservator Cuong Nguyen and Conservation Technician Andrew Ganem working on motors in their lab.
We have also been very fortunate to have Cuong and Andrew working with us for a little while. They're tackling some larger motors, which take longer to complete. Their help allows Conservation Specialist Mallory Bower and I to continue to work at the pace necessary to keep the project on target, while ensuring that as much of the collection as possible is treated. We greatly appreciate their help.
As always, this is only a small sampling of what we have been up to on the IMLS project. Please feel free to stop by our window at the back of the museum and see what we’re working on – there is always something interesting on our desks. Keep your eyes peeled for our next Facebook Live, as well. As we continue to move into 2017 and are fully into the second half of the project, we are excited to continue our work and continue keeping you updated
Louise Stewart Beck is former IMLS Project Conservator at The Henry Ford.
Additional Readings:
- Surprising Stories from IMLS Discoveries
- The Allegheny Articulated Steam Locomotive: Technology Pushed to the Limit
- #InnovationNation: Power & Energy
- The Ingersoll-Rand Diesel-Electric Locomotive: Boxy but Significant
power, electricity, by Louise Stewart Beck, collections care, conservation, IMLS grant
Behind the Scenes with IMLS: Recent Progress
 There has been a lot going on at The Henry Ford lately – our Beatles exhibit has just closed, the new Davidson-Gerson Modern Glass Gallery is soon to open, and the conservation department has been involved with those goings-on and more. Even though there’s a lot of change and activity, our IMLS-funded grant project to work on our electrical collections continues at a steady pace. As we approach the halfway point in the grant, we are also approaching 450 objects conserved – the halfway point of our 900-object goal!
There has been a lot going on at The Henry Ford lately – our Beatles exhibit has just closed, the new Davidson-Gerson Modern Glass Gallery is soon to open, and the conservation department has been involved with those goings-on and more. Even though there’s a lot of change and activity, our IMLS-funded grant project to work on our electrical collections continues at a steady pace. As we approach the halfway point in the grant, we are also approaching 450 objects conserved – the halfway point of our 900-object goal!
Conservation Specialist Mallory Bower and Senior Conservator Clara Deck clean objects in the Collections Storage Building.
We have been continuing to make regular trips to our Collections Storage Building (CSB) to select artifacts for inclusion in the grant; while we’re out there, we give them an initial clean, before bringing them into the museum to be fully conserved, then photographed and packed.

Collections Specialist Cayla Osgood brings down the dynamo on a forklift while Mallory “spots”, keeping a watchful eye for corners, overlapping edges, or any other potential issues.
We have recently brought our third “extra-large” object in from CSB, an Eickemeyer Dynamo. When choosing objects to bring in, we take into account the wants and needs of other departments of the museum, and we chose this object as there was some interest in it from the curatorial department. Since it was high up on a shelf, it had been a little while since they were able to inspect it up-close – there was a lot of excitement when we brought it in! Although it will not be going on display, it is now clean and accessible, and soon it will be digitized and available online. 
The Eickemeyer Dynamo, retrieved from storage (32.107.1)
The dynamo did not need an excessive amount of treatment, largely a brush/vacuum to remove storage dust, plus removal of a little copper corrosion on some of the fittings on the ends. (Want to read more about our “extra-large” objects? Check out our previous blog post!)

A circuit breaker with a marble base, during treatment (29.1333.292)
Although the “extra-large” objects have been focused on quite a bit in our blogs, most of what we do involves much smaller objects. There are so many different materials and types of objects, we have a lot of interesting challenges to work through. Something of particular note that we have come across a few times now is objects with marble bases, like this circuit breaker. The marble is frequently very dirty, with staining and significant accretions, and, as in this case, also cleans up fairly well! This “in progress” shot shows how different the object can look from when we get it out of the Collections Storage Building to when it’s clean and finished, ready to be digitized and packed.
So that’s where we stand currently, nearly halfway through our IMLS grant, working away on lots of electrical objects. Keep your eyes peeled for future blog posts with updates on our progress!
Louise Stewart Beck is former IMLS Project Conservator at The Henry Ford.
power, electricity, IMLS grant, #Behind The Scenes @ The Henry Ford, collections care, conservation, by Louise Stewart Beck
Behind the Scenes with IMLS: Cleaning Objects
 One of the main components of The Henry Ford’s IMLS-funded grant is the treatment of electrical objects coming out of storage. This largely involves cleaning the objects to remove dust, dirt, and corrosion products. Even though this may sound mundane, we come across drastic visual changes as well as some really interesting types of corrosion and deterioration, both of which we find really exciting.
One of the main components of The Henry Ford’s IMLS-funded grant is the treatment of electrical objects coming out of storage. This largely involves cleaning the objects to remove dust, dirt, and corrosion products. Even though this may sound mundane, we come across drastic visual changes as well as some really interesting types of corrosion and deterioration, both of which we find really exciting. An electrical drafting board during treatment (2016.0.1.28)
An electrical drafting board during treatment (2016.0.1.28)
Conservation specialist Mallory Bower had a great object recently which demonstrates how much dust we are seeing settled on some of the objects. We’re lucky that most of the dust is not terribly greasy, and thus comes off of things like paper with relative ease. That said, it’s still eye-opening how much can accumulate, and it definitely shows how much better off these objects will be in enclosed storage.
Before and after treatment images of a recording & alarm gauge (2016.0.1.46)
The recording and alarm gauge pictured above underwent a great visual transformation after cleaning, which you can see in its before-and-after-treatment photos. As a bonus, we also have an image of the material that likely caused the fogging of the glass in the first place! There are several hard rubber components within this object, which give off sulfurous corrosion products over time. We can see evidence of these in the reaction between the copper alloys nearby the rubber as well as in the fogging of the glass. The picture below shows where a copper screw was corroding within a rubber block – but that cylinder sticking up (see arrow) is all corrosion product, the metal was actually flush with the rubber surface. I saved this little cylinder of corrosion, in case we have the chance to do some testing in the future to determine its precise chemical composition.

Hard rubber in contact with copper alloys, causing corrosion which also fogged the glass (also 2016.0.1.46). Hard rubber corrosion on part of an object – note the screw heads and the base of the post.
Hard rubber corrosion on part of an object – note the screw heads and the base of the post.
This is another example of an object with hard rubber corrosion. In the photo, you can see it ‘growing’ up from the metal of the screws and the post – look carefully for the screw heads on the inside edges of the circular indentation. We’re encountering quite a lot of this in our day to day work, and though it’s satisfying to remove, but definitely an interesting problem to think about as well.
There are absolutely more types of dirt and corrosion that we remove, these are just two of the most drastic in terms of appearance and the visual changes that happen to the object when it comes through conservation.
We will be back with further updates on the status of our project, so stay tuned.
Louise Stewart Beck is Senior Conservator at The Henry Ford.
Michigan, Dearborn, 21st century, 2010s, IMLS grant, conservation, collections care, by Louise Stewart Beck, #Behind The Scenes @ The Henry Ford
 If you’ve ever walked by the conservation labs at the back of Henry Ford museum, you’ve probably seen the conservators at work on a variety of objects, of a variety of sizes. With a grant from the Institute of Museum and Library Services, we are primarily working on “bench-top” objects – which can be picked up and moved by hand. There are, however, a handful of extra-large objects that we have planned to work on over the course of the grant, including (but not limited to!) historically significant motors, electrostatic producers, and transformers. These objects are important within the electrical scope of the grant, and they need work to be stabilized and preserved for the future.
If you’ve ever walked by the conservation labs at the back of Henry Ford museum, you’ve probably seen the conservators at work on a variety of objects, of a variety of sizes. With a grant from the Institute of Museum and Library Services, we are primarily working on “bench-top” objects – which can be picked up and moved by hand. There are, however, a handful of extra-large objects that we have planned to work on over the course of the grant, including (but not limited to!) historically significant motors, electrostatic producers, and transformers. These objects are important within the electrical scope of the grant, and they need work to be stabilized and preserved for the future. Note that “extra-large” for us is a lot different than extra-large for the rest of the museum – the Allegheny is magnitudes larger than anything we are working with, for example! The “extra-large” objects that we are working on range up to 2 tons in weight, and require specialized equipment such as forklifts to move. We draw the line at artifacts requiring specialist rigging or outside contractors. These sorts of objects do bring their own issues – moving them from one place to another is difficult and requires careful planning, they require a good deal of space in the lab, and the treatments can take a significant length of time. We’re moving at a quick pace with the work on this grant, so taking two to three weeks just working on one object isn’t a good solution for us.

The first extra-large object we’ve grabbed, viewed top-down – a Sprague streetcar motor.
So how do we balance the amount of time it takes to treat very large objects with the need to keep up a pace in order to achieve completion goals? We’ve tackled this perennial problem in an interesting way. Since we don’t have an enormous number of extra-large objects to complete, we are allowing three months for the conservation of each. What this means practically is that we can bring the object into the lab, give it a space, and then as we have breaks between work on smaller objects, we can dedicate a few hours to it here and there. Breaking up the conservation work in this way has been very successful so far!The first object that we’ve treated in this way is a Sprague streetcar motor. This is a really interesting and important object, believed to have been used in Richmond, Virginia on the first major electric street railway system, and dating to the end of the 19th century.


Two of the coils on the motor before treatment.
In the image above are shown two of the coils on the motor before treatment – the textile covering was loose and dirty, and in some places the damage extended to the layer below the outer wrapping as well. The treatment for this object required not only cleaning, but repair to these areas of damage.

Their ‘tails’ have been rewound and reattached, and the dust and dirt have been removed. The area around the coils has also been cleaned and the wire wrappings have been tidied. The engine overall is nearing completion, but does have some areas that still need cleaning. It’s been great to have it as a project we can come back to for small spurts of time, which is exactly what we were hoping for our extra-large object treatment plan.
Louise Stewart Beck is IMLS Conservator at The Henry Ford.
#Behind The Scenes @ The Henry Ford, collections care, conservation, by Louise Stewart Beck, IMLS grant

Conservation specialist Mallory Bower and collections specialist Jake Hildebrandt removing objects from shelving in the Collections Storage Building. The Henry Ford has recently embarked on a new adventure, thanks to a grant from the Institute of Museum and Library Services (IMLS) which allows us to spend time working on the electrical collections currently housed in the Collections Storage Building. This is The Henry Ford’s second IMLS grant dedicated to improving the storage of and access to collections. The first grant focused on communications technology, and was completed over two years, ending in October 2015. At the end of that grant, more than 1000 communications-related objects were conserved, catalogued, digitized, and stored, marking a huge improvement in the state of the collections and their accessibility. We have similar goals for this grant, as we aim to complete 900+ objects by October 2017.
The Henry Ford has recently embarked on a new adventure, thanks to a grant from the Institute of Museum and Library Services (IMLS) which allows us to spend time working on the electrical collections currently housed in the Collections Storage Building. This is The Henry Ford’s second IMLS grant dedicated to improving the storage of and access to collections. The first grant focused on communications technology, and was completed over two years, ending in October 2015. At the end of that grant, more than 1000 communications-related objects were conserved, catalogued, digitized, and stored, marking a huge improvement in the state of the collections and their accessibility. We have similar goals for this grant, as we aim to complete 900+ objects by October 2017.
digitization, electricity, power, by Louise Stewart Beck, conservation, collections care, IMLS grant, #Behind The Scenes @ The Henry Ford

